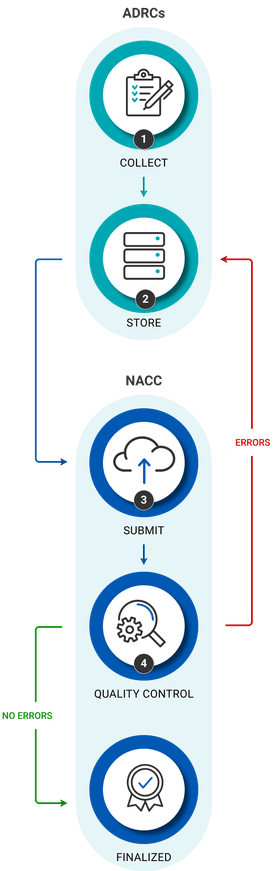
Data submission overview
1. Collect clinical data
Clinicians at ADRCs collect clinical data using NACC forms. All ADRCs administer the UDS, though there are additional forms that should be administered when possible, including the FTLD, LBD, and neuropathology modules. For a better understanding of data being collected, look through NACC forms and documentation which includes several types of documents:
- Forms packets: The actual clinical forms that will be administered to patients.
- Guidebooks: Instructions for clinicians describing how to administer the packets.
- Data element dictionaries (DEDs): Technical details about form variables which can help you set up data storage.
- Data templates: Technical details about the data structure expected by the NACC submission system.
ADRCs are welcome to rebuild clinical forms in their EDC software of choice, following the NACC e-forms guidelines.
2. Store data locally
ADRCs either build a database or leverage data collection tools with built-in storage. There is no standard tool prescribed by NACC. The data capture tool most commonly used among ADRCs is REDCap, and the ADRC community has shared their REDCap configurations which would be a great place to start for any ADRCs considering adoption of this tool.
3. Submit data to NACC
When ready to submit data, ADRCs can reach out to nacchelp@uw.edu. NACC will create user accounts allowing access to the data submission system which supports:
- Bulk submission: By uploading csv files that meets the NACC expected format, a large amount of data can be submitted at one time. For ADRCs using REDCap, 1Florida ADRC has created a great open-source tool called the NACCulator which converts REDCap data into NACC formats, simplifying the bulk submission process.
- Data entry: A web-based interface allows data entry form-by-form.
- Mixed Protocol image submission: Sites can submit non-SCAN compliant MRI and PET images directly to NACC – Learn more here.
4. Run QC
After submission, data need to pass a full battery of QC "error checks" which can be kicked off from the NACC submission system. A list of errors is displayed, ADRCs fix the errors, re-submit data, and re-run error checks. This process is repeated until no errors are found, at which point the data is considered "finalized" and is ready for distribution to the research community.